Explore our professional guide to Potentiometric Titration procedures in accordance with IP, BP, USP, and Ph. Eur. Ensure precision in your testing with our detailed article and standard test procedure
Potentiometric Titration Procedure
1.0 Objective
-
- The objective of this General Test Procedure (GTP) is to describe the procedure to be used for performing of potentiometric titration.
2.0 Principle/Reaction
-
- It is a quantitative measuring technique used to find the variation of the potential difference between 2 electrodes (either one indicator electrode and one reference electrode or 2 indicator electrodes) immersed in the solution to be examined as a function of the quantity of titrant added.
3.0 Requirements: as per USP/BP/Ph. Eur./IP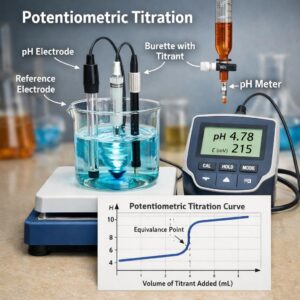
-
- Electrode
4.0 Potentiometric Titration Procedure as per USP
-
- The simplest and most convenient method by which the equivalence point, i.e., the point at which the stoichiometric analytical reaction is complete, may be determined is with the use of indicators.
-
- These chemical substances, usually colored, respond to changes in solution conditions before and after the equivalence point by exhibiting color changes that may be taken visually as the endpoint, a reliable estimate of the equivalence point.
-
-
A useful method of endpoint determination results from the use of electrochemical measurements.
-
-
- If an indicator electrode, sensitive to the concentration of the species undergoing titrimetric reaction, and a reference electrode, whose potential is insensitive to any dissolved species, are immersed in the titrate to form a galvanic cell, the potential difference between the electrodes may be sensed by a pH meter and used to follow the course of the reaction.
-
- Where such a series of measurements is plotted correctly (i.e., for an acid-base titration, pH versus mL of titrant added; for a precipitimetric, complexometric, or oxidation-reduction titration, mV versus mL of titrant added), a sigmoid curve results with a rapidly changing portion (the “ break”) in the vicinity of the equivalence point.
-
- The midpoint of this linear vertical portion or the inflection point may be taken as the endpoint.
-
- The equivalence point may also be determined mathematically without plotting a curve.
-
- However, it should be noted that in asymmetrical reactions, which are reactions in which the number of anions reacting is not the same as the number of cations reacting, the endpoint as defined by the inflection of the titration curve does not occur exactly at the stoichiometric equivalence point.
-
- Thus, potentiometric endpoint detection by this method is not suitable in the case of asymmetric reactions, examples of which are the precipitation reaction,
2Ag + + CrO4–2
and the oxidation-reduction reaction,
5Fe +2 + MnO4–.
-
-
All acid-base reactions, however, are symmetrical.
-
-
- Thus, potentiometric endpoint detection may be employed in acid-base titrations and in other titrations involving symmetrical reversible reactions where an indicator is specified, unless otherwise directed in the individual monograph.
-
- Two types of automatic electrometric titrators are available.
-
- The first is one that carries out titrant addition automatically and records the electrode potential differences during the course of titration as the expected sigmoid curve.
-
- In the second type, titrant addition is performed automatically until a preset potential or pH, representing the endpoint, is reached, at which point the titrant addition ceases.
-
- Several acceptable electrode systems for potentiometric titrations are summarized in Table.
Potentiometric Titration Electrode Systems
| Titration | Indicating Electrode | Equation 1 | Reference Electrode | Applicability |
| Acid-base | Glass | E=k + 0.0591 pH | Calomel or silver–silver Chloride | Titration of acids and bases |
| Precipitimetric (silver) | Silver | E=E+0.0591 log [Ag+] | Calomel (with potassium nitrate salt bridge) | Titration with or of silver involving halides or thiocyanate |
| Complexometric | Mercury– mercury(II) | E=E + 0.0296 (log k ′ pM) | Calomel | Titration of various metals (M), e.g., Mg +2, Ca Al +3 , Bi +3, with EDTA |
| Oxidation – Reduction | Platinum | E=E + (0.0591/n) × log [ox]/[red] | Calomel or silver–silver chloride | Titrations with arsenite, cerate, bromine, dichromate, exacyonoferrate (III), iodate, nitrite, permanganate, thiosulfate |
-
- Note: Appropriate form of Nernst equation describing the indicating electrode system: k= glass electrode constant; k′=constant derived from Hg–Hg(II)– EDTA equilibrium; M =any metal undergoing EDTA titration; [ox] and [red] from the equation, ox + ne → red. Listing is repesentative but not exhaustive.
-
-
Blank Corrections:
-
-
- As previously noted, the endpoint determined in a titrimetric assay is an estimate of reaction equivalence point.
-
- The validity of this estimate depends upon, among other factors, the nature titrate constituents and the concentration of the titrant.
-
- An appropriate blank correction is employed in titrimetric assays to enhance the reliability of the endpoint determination. Such a blank correction is usually obtained by means of a residual blank titration, wherein the required procedure is repeated in every detail except that the substance being assayed is omitted.
-
- In such instances, the actual volume of titrant equivalent to the substance being assayed is the difference between the volume consumed in the residual blank titration and that consumed in the titration with the substance present.
-
- The corrected volume so obtained is used calculating the quantity of the substance being titrated, in the same manner as prescribed under Residual Titrations.
-
- Where potentiometric endpoint detection is employed, the blank correction is usually negligible.
5.0 Potentiometric Titration Procedure as per BP / Ph. Eur
-
- The solvent mixture is neutralized, if necessary, before dissolution of the substance to be examined.
-
- Plot a graph of the variation of potential difference as a function of the quantity of the titrant added, continuing the addition of the titrant beyond the presumed equivalence point.
-
- The end-point corresponds to a sharp variation of potential difference.
-
-
Potentiometric Titration Apparatus:
-
-
- The apparatus used comprises a millivolt meter commercial autotitrator instrument may be used and are operated in accordance with the manufacturer’s instructions using electrode recommended for the type of titration described.
-
- The indicator electrode to be used depends upon the substance to be determined and may be a metal electrode (Platinum, gold, silver).
-
- For acid-base titrations, a glass-silver chloride electrode combination is generally used.
-
-
Potentiometric Titration Method:
-
-
- Prepare the sample solutions as described.
-
- Add the titrant in suitable aliquots paying particular attention to the rate of addition and the volume increments near the end-point.
-
- Continue the titration beyond this point to allow a clear detection of end-point.
-
- The end-point of the titration is reached when the maximum change in potential occurs in a plot of potential versus volume of titrant, and is expressed as the corresponding volume of titrant.
-
- Record the first or second derivatives curve can facilitate the determination of the end-point.
-
- In potentiometric titrations of weak acid or weak base using non-aqueous solvents, if necessary, either carry out blank determination pre-neutralize the solvent mixture.
-
- Where it is impracticable to use potentiometric detection for this purpose, the solvent mixture can be pre-neutralized by titration using suitable indicator.
6.0 Potentiometric Titration Procedure as per IP
-
- Plot a graph of the variations of potential difference versus the volume of the titrant added, continuing the addition of the titrant beyond the presumed equivalence point.
-
- The end-point corresponds to a sharp variation of potential difference.
-
- Perform a blank titration by repeating the procedure in the same manner omitting the substance under examination.
-
- The actual volume of titrant equivalent to the substance under examination is the difference between the volume consumed in the blank titration and that consumed in the titration with the volume consumed in the blank titration and that consumed in the titration with the substance under examination.

